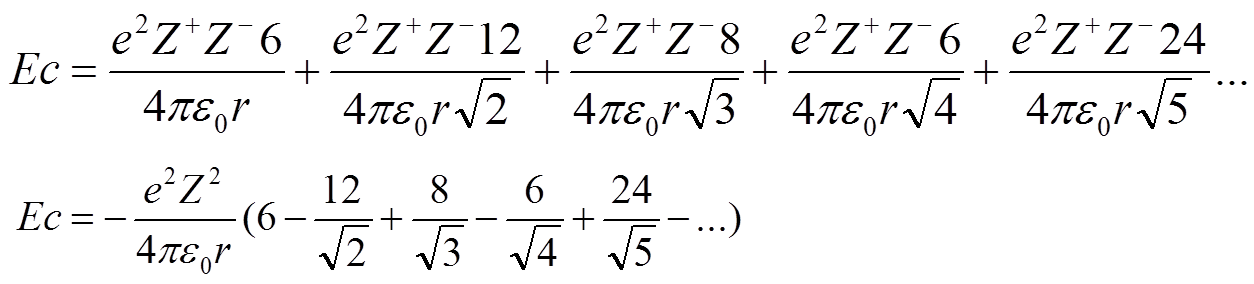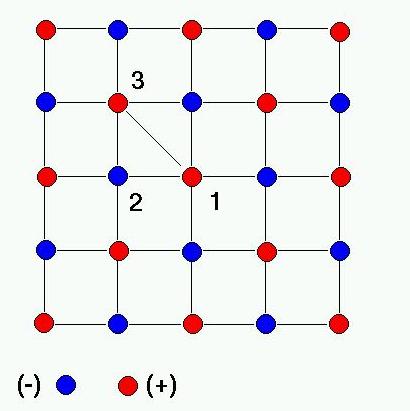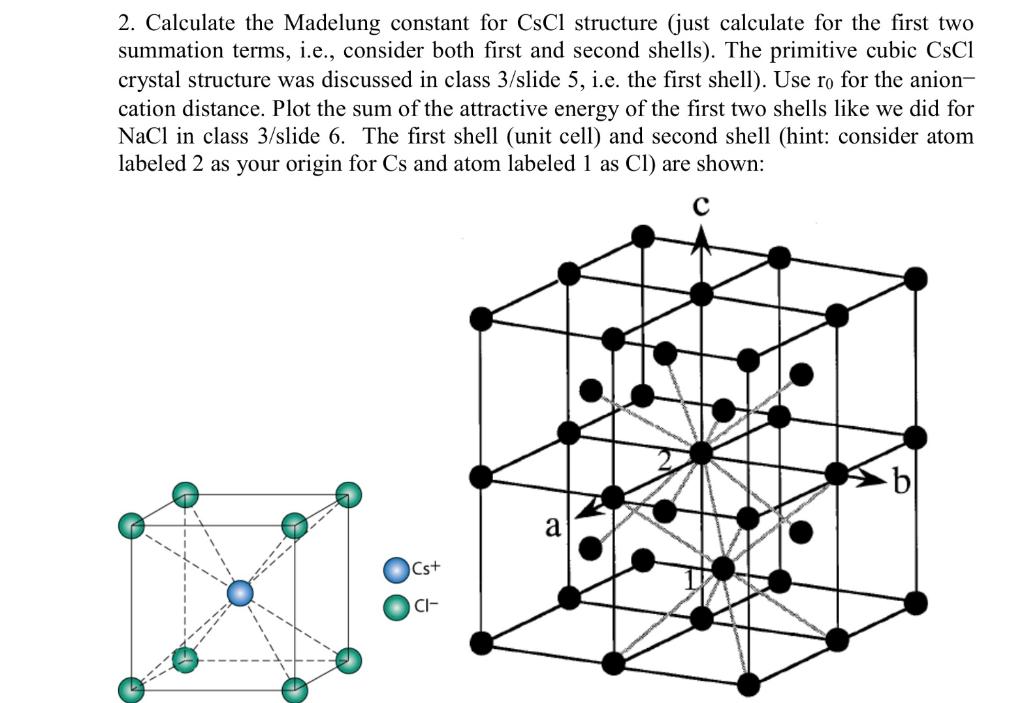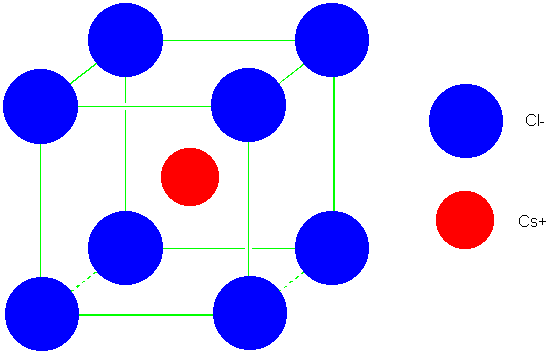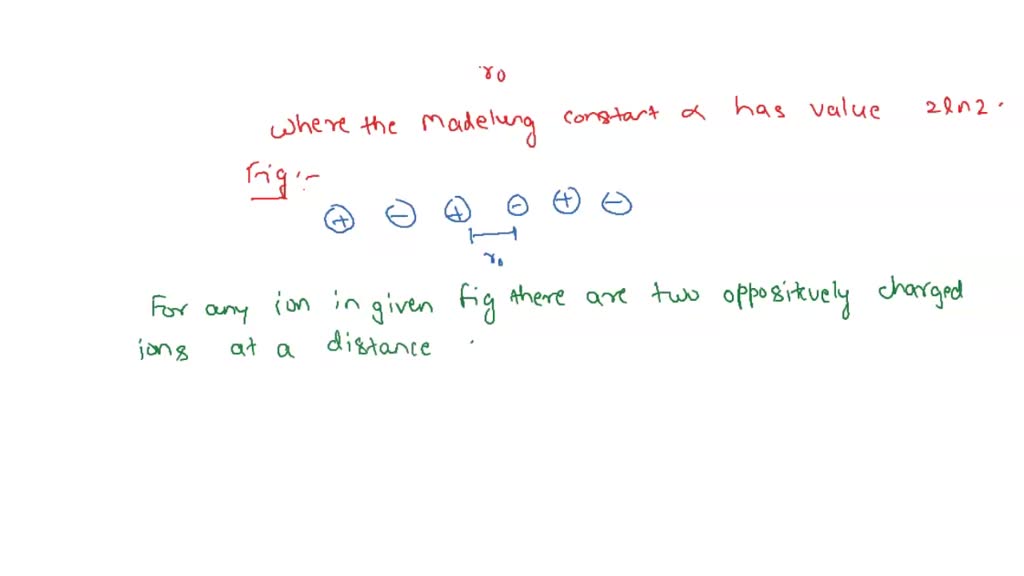
SOLVED: '3) Ionic bonding: The Madelung constant for three-dimensional crystal of NaCl was presented Derive the Madelung constant analytically for a one-dimensional chain of NaCl,as shown in Figure 2.3 . Calculate the
Show that the Madelung constant for a one-dimensional array of ions of alternating sign with - Sarthaks eConnect | Largest Online Education Community

Calculate the lattice energy of sodium chloride crystal from the following data: Born exponent, n=8, Madelung constant for NaCl=1.748, Ionic radius of Na^(+)=0.95 Å, Ionic radius of Cl^(-)=1.81Å.

The calculation of the madelung constant for NaCl structure using the method of images - Nafari - 1987 - physica status solidi (a) - Wiley Online Library

The calculation of the Madelung constant of a “generalized” sodium chloride lattice - Transactions of the Faraday Society (RSC Publishing)